Fed State Simulated Intestinal Fluid (FeSSIF) Dried Powder BZ552
Product Code: BZ552
Manufacturer: Biochemazone
Available Pack Sizes for Making: 100 mL, 200 mL, 500 mL, 1000 mL
Custom Bulk Quantities Available
Product Overview
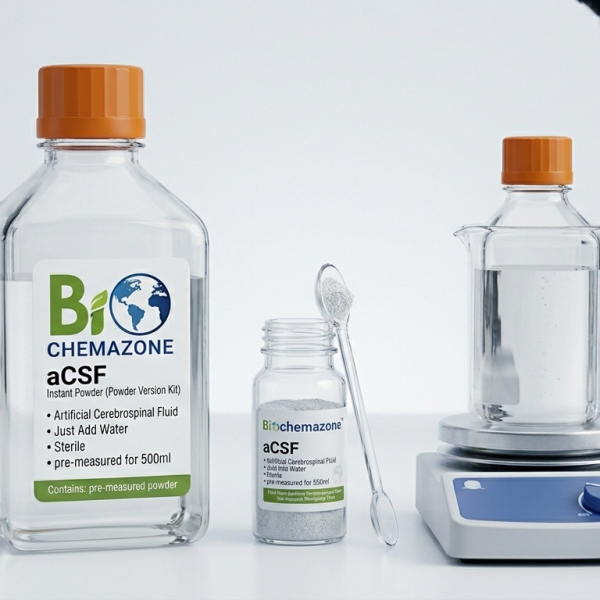
Biochemazone Fed State Simulated Intestinal Fluid (FeSSIF) Dried Powder BZ552 is a laboratory-formulated powdered medium designed to simulate the physiological composition and biorelevant conditions of the human small intestine under fed-state conditions. This dry-format formulation allows convenient storage, transportation, and preparation of fresh simulated intestinal fluid prior to experimental use.
The formulation is specifically developed to mimic the intestinal environment encountered in the upper gastrointestinal tract after food intake, including elevated bile salt content, phospholipid composition, buffering capacity, digestive surface activity, and physiological fed-state pH conditions. This makes it highly suitable for pharmaceutical dissolution studies, oral drug delivery research, bioavailability assessment, and biopharmaceutical testing applications.
Prepared using high-purity analytical-grade components, this standardized dried powder formulation provides excellent reproducibility and consistency for in vitro and preclinical studies. Upon reconstitution with purified water, the solution simulates intestinal fluid conditions relevant to solubility enhancement, dissolution behavior, permeability assessment, lipid-based formulation testing, and oral dosage form performance under fed-state conditions.
BZ552 is widely used in oral drug formulation development, dissolution and release testing, bioaccessibility studies, lipid formulation evaluation, and biorelevant media testing. The formulation contains no preservatives and may also be customized for specific pH ranges, bile salt concentrations, surfactant systems, or specialized gastrointestinal simulation requirements depending on research applications.
Key Features & Benefits
✅ Simulated Fed-State Intestinal Environment – Designed to mimic physiological intestinal fluid conditions after food intake
✅ Dried Powder Format – Convenient storage and preparation of fresh media
✅ Preservative-Free Formulation – Suitable for sensitive research applications
✅ Biorelevant Composition – Mimics fed-state intestinal bile salt and phospholipid environment
✅ Physiological pH Simulation – Suitable for gastrointestinal dissolution studies
✅ Reproducible & Standardized – Ideal for comparative laboratory applications
✅ High-Purity Components – Prepared using analytical-grade raw materials
✅ Freshly Reconstituted Medium – Enhances experimental consistency and stability
✅ Ready-to-Prepare Formulation – Simple one-step reconstitution for laboratory use
Applications
🧪 Pharmaceutical & Drug Delivery Research
- Oral drug dissolution and release studies
• Solubility and bioavailability enhancement testing
• Lipid-based drug delivery system evaluation
🐀 Preclinical & Biomedical Studies
- Gastrointestinal environment simulation
• Permeability and absorption assessment
• Controlled-release oral formulation studies
🔬 Biopharmaceutical & Analytical Research
- Biorelevant dissolution testing
• Supersaturation and precipitation studies
• Comparative formulation performance evaluation
🧬 Drug Development & Formulation Studies
- pH-dependent oral formulation assessment
• BCS classification support studies
• Amorphous and poorly soluble drug evaluation
Typical Properties
- Appearance: Off-white to white free-flowing powder
• Reconstituted Appearance: Clear to slightly opalescent solution
• pH (after reconstitution): Physiological fed-state intestinal pH range
• Solubility: Water soluble upon reconstitution
• Composition: Buffered intestinal electrolyte, bile salt, and phospholipid formulation, Pathogen Free
• Preservatives: None
• Buffering Capacity: Physiologically balanced intestinal buffering system
• Format: Dried powder preparation
Storage & Stability
- Storage Temperature: 2–30 °C recommended
• Shelf Life: 12–24 months (sealed container)
• Shipping: Stable at room temperature
Customization Options
- pH adjustment for specific gastrointestinal studies
• Customized bile salt and phospholipid concentrations
• Surfactant and lipid composition modifications
• Fasted-state simulation modifications available upon request
• Species-specific or study-specific intestinal simulation customization
Ordering Information
BZ552 is available in standard laboratory sizes or OEM/bulk supply formats.
📄 MSDS, Technical Datasheet, and Certificate of Analysis (CoA) available upon request
📧 Email: info@biochemazone.com
🌐 Website: Biochemazone
⚠️ For research, education, and in vitro testing only. Not for human or animal use